- Blog
- Image viewer for windows 8
- Intel iris pro 1536 mb nvidia equivalent
- Vetri dmg for dogs
- Mc afee online
- Arcgis 10-6 inserted image not appearing
- Como emular o eutron
- Ningbo wincar auto accessories legitimate
- What is the rhema word of god
- Transcad data miami
- Help using es file explorer pro
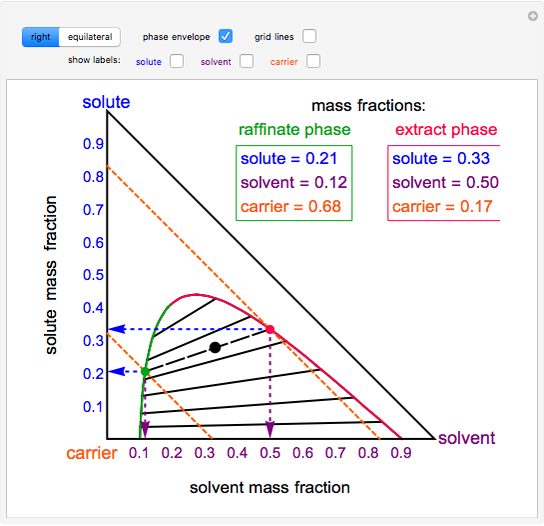
- Two phase region ternary diagram
- Airfoil generator software
- Yu gi oh gx sub indo
- Free pagemaker alternative
- Pocket girlfriend ipa
Maximum solubility of Ag in Cu occurs at 779☌ and is 8%. Below line CEG (779☌), there is only partial solubility of Ag in Cu (α phase) and Cu in Ag (β-phase). (α + β) phase constitutes of pure copper and pure silver. It is rich in Ag and Cu is present as solute and again has FCC structure. It is rich in copper and Ag is present as solute and has FCC structure. So three phase α, β, L will be there at eutectic point. Eutectic means in latin word Eu means: nice & tectic means: melting.į = 0, So by Gibb’s law O + P = 2 + 1 P = 3. Here a eutectic point is defined, where degree of freedoms are zero.

The material which are completely soluble in liquid state but partially soluble in solid state (eutectic phase diagram).īinary Eutectic system consists of three phase: Phase diagrams give three kinds of informations: Melting or freezing occurs over a range of temperatures between solids and liquidus lines. Solidus and liquidus lines intersect at two extremities (composition) point of intersection represents the melting point of pure metal (Cu and Ni). This is a mushy zone where both Cu and Ni are present in varying compositions. (α + L)- Phase between liquidus line and solidus line. Liquid (L)- It is a homogenous liquid solution composed of both Cu and Ni. At temperature below 1085☌ Cu and Ni are mutually soluble in each other in solid state for all composition as both have same FCC crystal structure and nearly identical atomic radii and electronegativity. This phase is a substitutional solid solution of Cu and Ni atoms and having FCC structures. When process is carried out below the recrystallization temperature (0.3T m to 0.5T m) these are known as cold working and above of it is termed as hot working. So, in this case Gibb’s rule converts intoīasically, when the material transform from one phase to another phase two type of process occur: If the entire length of the tie line mo is taken to represent 100% or the total weight of the two phases present at temperature T, the lever rule may be expressed mathematically as:įor such phase diagram, there is one degree of freedom. In the previous figure, the vertical line representing the alloy 20B, divides the horizontal tie line into two parts ‘mn’ and ‘no’. The relative length of the lever arms multiplied by the amounts of the phase present must balance. The point where the vertical line intersects the horizontal line may be considered as the fulcrum of a lever system. The vertical line will divide the horizontal line into two parts whose lengths are inversely proportional to the amount of phase present. To determine the relative amount of the two phase in equilibrium at any specified temperature in a two phase region, draw a vertical line representing the alloy and a horizontal temperature line to the boundaries of the field. Similarly, point 0, when dropped to the base line, will give the composition of the other phase constituting the mixture, in this case liquid solution of composition 74A-26B. In this case, the phase is a solid solution a of composition 90A-10B. Point ‘m’, the intersection of the tie line with the solidus line, when dropped to the base line, gives the composition of the phase that exists at that boundary. These points of intersection are dropped to the base line, and the composition is read directly, e.g., in the below figure ‘mo’ tie line to the boundaries of the field. To determine the actual chemical composition of the phase of an alloy, in equilibrium at any specified temperature in a two-phase region, draw a horizontal temperature line (called a tie line) to the boundaries of the field.
- Blog
- Image viewer for windows 8
- Intel iris pro 1536 mb nvidia equivalent
- Vetri dmg for dogs
- Mc afee online
- Arcgis 10-6 inserted image not appearing
- Como emular o eutron
- Ningbo wincar auto accessories legitimate
- What is the rhema word of god
- Transcad data miami
- Help using es file explorer pro
- Two phase region ternary diagram
- Airfoil generator software
- Yu gi oh gx sub indo
- Free pagemaker alternative
- Pocket girlfriend ipa
